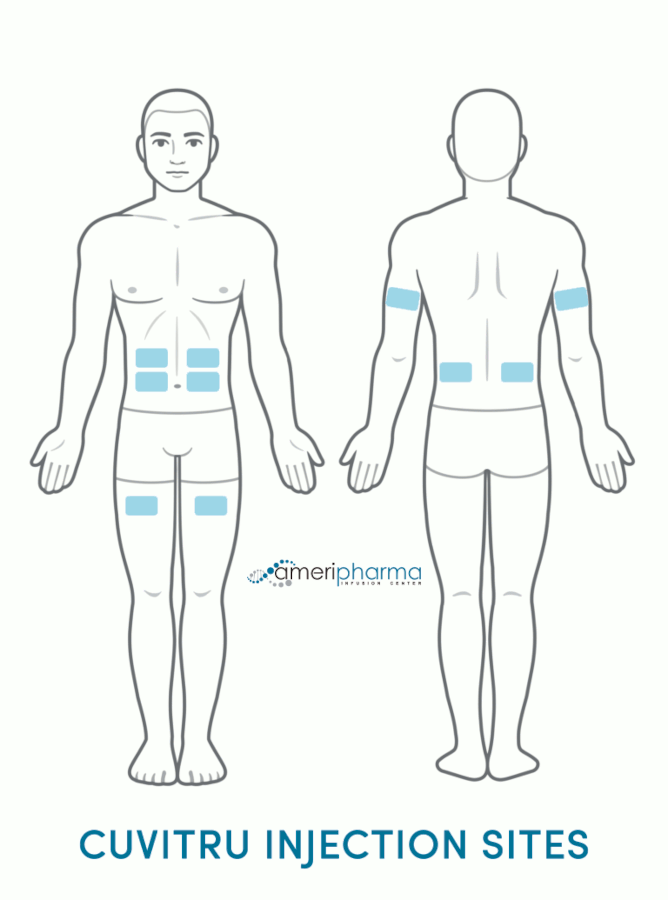
Cuvitru (immune globulin) is a medication derived from human plasma that is administered to patients with antibody deficiencies to help protect against infections. Plasma is a vital component of blood, providing its liquid base and containing various elements, such as water, blood clotting factors, plasma proteins, electrolytes, and immunoglobulins.
Cuvitru belongs to the immune globulins drug class and is FDA-approved for use as a monotherapy or add-on therapy with other medications.
Cuvitru Therapy and Indications
Cuvitru is an immunoglobulin that is administered by subcutaneous (under the skin) infusion. It works by replacing antibodies that are either not working correctly or missing in the human body, thereby helping to fight off infections. The doses aim to raise extremely low blood immunoglobulin levels to the required levels.
Cuvitru is recommended for several medical conditions, including:
- Primary Humoral Immunodeficiency (PI) in adults and children aged two years and above who have a congenital inability to produce antibodies.
- Patients with blood cancer experiencing low antibody production and recurrent infections when prophylactic antibiotics are not an option.
- Individuals with bone marrow cancer and those who have insufficient antibody production along with frequent infections.
- Patients who exhibit reduced antibody levels after receiving bone marrow cell transplants from a different person.
Side Effects
Patients receiving Cuvitru treatment may experience a range of side effects and symptoms that require prompt medical care, such as serious allergic reactions, urination problems, high or low blood pressure, respiratory infections and inflammation, aseptic meningitis, and injection-site reactions (e.g., indented skin, warmth).
Listed below are some of the possible side effects from using Cuvitru:
Allergic Reactions:
- Skin rash
- Elevated temperature
- Urticaria
- Itchiness
- Difficulty breathing
- Chest constriction
- Breathing issues
- Shortness of breath
- Swollen, red skin
- Facial and lip swelling
Symptoms That Require Prompt Medical Care:
- High fever
- Chills
- Abdominal distension
- Hematoma
- Edema
- Convulsions
- Abdominal discomfort
- Disorientation
- Fatigue
- Discolored urine
- Throat pain
- Mood Swings
- Jaundice
- Excessive sweating
- Irregular heartbeat
- Visual disturbances
- Speech issues
Urinary Issues:
- Weight gain
- Hematuria
- Increased urination
- Trouble urinating
Blood Pressure Changes:
- Lightheadedness
- Headache
- Impaired vision
Respiratory Infections and Inflammation:
- Nasal inflammation
- Sinus infection
- Bronchial asthma
Aseptic Meningitis (May Need Immediate Medical Attention):
- High fever
- Cold sensations
- Vomiting
- Neck stiffness
- Headache
- Stomach upset
- Sensitivity to light
Injection Site Reactions:
- Local itchiness
- Slight discomfort
- Skin redness
Note: Cuvitru injection-site reactions typically resolve within hours and usually do not require immediate treatment. Additionally, these reactions become less likely to happen as the body gets used to the medication.
Risk Factors for Blood Clots
Cuvitru has a Black Box Warning because of the possibility of blood clots. Patients with certain risk factors, such as estrogen use, advanced age, a sedentary lifestyle, hypercoagulable conditions, or a history of previous blood clots, are at a higher risk.
In order to minimize or mitigate the risk of blood clots, the manufacturer advises administering the infusion at the lowest possible dose and infusion rate. It is essential to monitor for any signs and symptoms throughout the treatment process.
Dosing Information
Cuvitru is injected under the skin (subcutaneously) by a qualified healthcare professional.
Dosing varies from person to person because it can depend on many things, such as body weight, medical history, blood IgG levels, current conditions, and clinical response to the treatment.
Considering these factors, the patient (either a child or an adult) may be prescribed an initial loading dose ranging from 1.0 to 2.5 ml/kg of body weight, divided over several consecutive days.
After this initial loading dose, patients will begin to receive Cuvitru infusions at consistent intervals, which can range from daily to once every two weeks.
Following the loading dose, the total monthly dose typically varies between 1.5 and 5.0 ml/kg of body weight. To attain the desired clinical outcome and serum IgG levels, the physician may modify the dose over time.
For those transitioning from a different IVIG brand, the Cuvitru dosage will be determined by the previous IVIG dosage and adjusted based on the quantity and frequency of infusions.
Never change the dosage or the dosing interval (how often you receive the medication) without discussing with your prescriber first.
In case of a missed dose, contact your healthcare provider right away for further instructions. Refrain from doubling the dose to compensate.
Cuvitru Uses
Cuvitru is used to treat various conditions, including primary immunodeficiency diseases (PI), idiopathic thrombocytopenic purpura (ITP), chronic inflammatory demyelinating polyneuropathy (CIDP), chronic immune thrombocytopenic purpura, and secondary humoral immunodeficiency diseases.
Cuvitru is also used to boost muscle strength in patients with multifocal motor neuropathy (MMN).
Cuvitru Cost and Copay Assistance
The cost of Cuvitru can depend on several factors, including:
- The pharmacy providing the medication
- The prescribed dose
- The patient’s insurance coverage
Cuvitru is provided in single-dose glass vials with typical volumes of 5 ml, 10 ml, 20 ml, 40 ml, and 50 ml protein per vial.
The base price shown on various pharmacy websites is determined by the manufacturer, Takeda Pharmaceutical, and does not include shipping fees or local taxes.
The cost of Cuvitru at different pharmacies is approximately $246 (around $49.50 per unit) for a 5 ml supply. Consequently, the total cost for a 50 ml supply is roughly $2,460 [1].
Pharmaceutical firms often establish various patient assistance programs (PAPs) to provide free or reduced-price medications to individuals who cannot afford their prescriptions.
One such initiative is Takeda’s OnePath Co-Pay Assistance Program, which supports patients prescribed Cuvitru [2]. To be eligible for this program, patients must have commercial insurance.
This program helps with certain out-of-pocket treatment expenses and aids in navigating the health insurance process. It also offers in-house training from qualified personnel and access to educational and support services.
Additionally, the HelloCUVITRU program is a complimentary trial that covers the complete cost of the initial four Cuvitru infusions, as well as the necessary administration supplies [3]. To qualify for this program, patients must be at least 2 years old, have not previously participated in the program, and are not currently using the medication.
Conclusion
Cuvitru is a crucial medication for patients with immunodeficiency conditions and helps protect against infections by replacing antibodies in the body. It is essential for patients to consult with their healthcare professional to determine the appropriate dosing regimen and to monitor for potential side effects. As the cost of Cuvitru can be high, it is recommended to explore available assistance programs to help cover treatment costs. By following the prescribed treatment plan and maintaining open communication with healthcare providers, patients can effectively manage their immunodeficiency conditions and improve their overall quality of life.
References:
1. https://www.drugs.com/price-guide/cuvitru
2. https://www.takeda.com/en-ca/what-we-do/helping-canadian-patients/onepath-assistance-program/
3. https://www.cuvitru.com/hello-cuvitru